High-Throughput CRISPR Screening in Primary Cells with Mechanoporation on Galaxy-i
Who this is for:
- Cell therapy developers scaling primary cell engineering workflows to screening campaigns
- Immunology and gene editing labs performing high-throughput optimization of guides in primary immune cells
- Assay developers looking to run high throughput crispr screens on primary cells
Opportunties
- Generate large matrixed CRISPR datasets (multiple guides × donors × cargos) with consistent delivery, enabling cleaner comparisons and better hit-calling.
- Support multi-component editing (RNP + ssDNA, dual-guide systems, large RNP complexes) across plates without the performance drop-offs common in primary cells.
- Per well costs <$1 for 96 well plates and less than $0.25 per well for 384 well plates.
- Confidently scale exploratory Gateway conditions to full plates, knowing wells behave similarly.
- Enable repeatable screening runs across donors, making follow-up validation datasets more reliable.
Results Obtained
- 87% primary T cell average viability maintained across all 96 wells through 100+ sequential dispenses
- 74-80% average delivery efficiency for both gene knockout (B2M-KO) and a model small molecule (dextran)—proving that complex cargo delivery to primary cells can scale without compromise
Plate Based Screening of B2M RNP + Dextran Delivery to Unstimulated Primary Human T Cells on Certus Flex
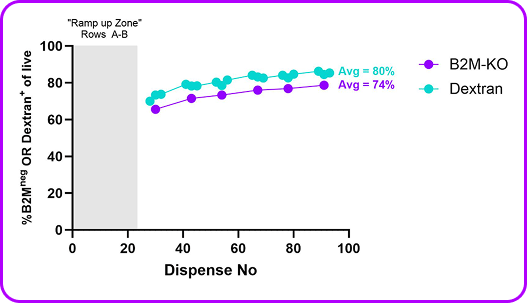
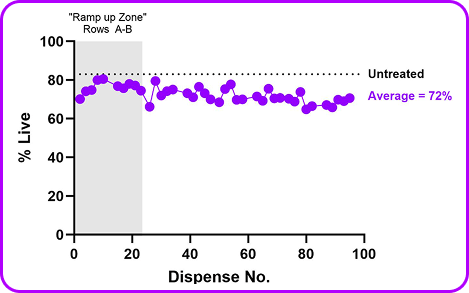
B2M RNP was pre-loaded into a 96 well plate and unstimulated primary human T cells were mechanoporated into each well. Viability and editing efficiency were measured by flow on day 3 post-mechanoporation. Consistent and high delivery & viability was seen across the plate validating this as a screening method.
%203.png)
Rewatch the Webinar: High-Throughput Mechanoporation Using Certus Flex Integration
Additional High Throughput Data
Broad Cell Type & Cargo HT Delivery Galaxy-i (PDII)
Additional screening workflows have been run on the HighRes PDII across a wide range of cargo & cell type. You can learn more about the PDII integration here!

Consistent Delivery Across Plates: In experiments with HeLa cells across five 96-well plates, we demonstrated consistent delivery across all five plates. This consistent performance was demonstrated across cargos using 3kDa dextran, labeled oligo, and IgG antibody.
.png?width=1200&length=1200&name=Group%2049%20(3).png)
A) PD II In-line Integration Enables Scalable and Consistent High-Throughput delivery. HeLa cells were mechanoporated ("boosted") using either the Portal R&D system (blue) or the Portal cartridge integrated into the PDII dispenser into wells of 5, 96-well plates containing 3kDa fluorescently labeled dextran at 0.1mg/ml. Intracellular delivery to live cells was measured by flow cytometry.
.png?width=1200&length=1200&name=Group%2047%20(1).png)
.png?width=1200&length=1200&name=Group%2048%20(3).png)
Broad Cell Type Compatibility: The technology isn't limited to cell lines. We demonstrated successful delivery in HEK293 cells (>60% oligo delivery, >50% antibody delivery) and human peripheral blood mononuclear cells (PBMCs) (>70% dextran delivery). This versatility is crucial for drug discovery programs that need to validate findings across multiple cell types.
.png?width=1200&length=1200&name=Group%2050%20(1).png)
.png?width=1200&length=1200&name=Group%2051%20(2).png)
.png?width=1200&length=1200&name=Group%2052%20(2).png)
Complex Cargo Delivery: Perhaps most exciting, we showed successful delivery of GFP mRNA to HeLa cells, maintaining high cell viability (>90%) while achieving >65% transfection efficiency. This opens doors for screening applications involving gene editing tools, regulatory RNAs, and other complex biological cargos.
.png?width=1200&length=1200&name=Group%2053%20(1).png)
.png?width=1200&length=1200&name=Group%2054%20(1).png)


Applications Unlocked by HT Mechanoporation
Impermeable Peptides
Transform your drug discovery pipeline with intracellular impermeable peptide delivery that maintains native cellular context. Learn more here!
DELs
Transform your drug discovery pipeline with intracellular DEL delivery that maintains native cellular context. Our latest application note demonstrates robust delivery of fluorescently-tagged DEL libraries to iPSCs, HeLa cells, and PBMCs with no bias in compound diversity. Key benefits include higher probability that identified hits will continue to bind targets in their desired cellular context, and the ability to screen against intracellular targets in their native environment. Learn more about direct-to-biology dels.
Intracellular Protein Detection
Detect intracellular proteins in real-time in living cells. Mechanoporation delivers LgBiT directly into living cells, enabling time-resolved protein level analysis assays without lysing your cells. See the results here!
Circular RNA
Achieve 5+ days of gene expression in primary cells without genome editing. Mechanoporation delivers unmodified circular RNA for sustained protein production and enhanced CAR-T performance, no genetic modifications required. See the results
What This Means for Drug Discovery
This integration represents more than just a technical achievement, it's a paradigm shift toward more biologically relevant screening. By enabling direct-to-biology live cell assays at high-throughput scale, we're helping researchers:
- Validate targets earlier: Test compound activity directly in the cellular environment where it matters most
- Reduce late-stage failures: Identify potential issues before investing heavily in optimization
- Explore new chemical space: Screen compound classes that were previously impossible to test in cells
- Work with primary cells: Conduct more disease-relevant studies using patient-derived cells
Looking Forward: The Future of Live Cell Screening
The pharmaceutical industry is increasingly recognizing that better early-stage screening leads to better drugs reaching patients. Our partnership with HighRes Biosolutions represents a significant step toward making truly biological screening the standard rather than the exception.
As we continue to refine this technology and expand its applications, we're excited about the possibilities ahead. From screening massive compound libraries directly in disease-relevant cell models to enabling novel therapeutic modalities that require intracellular delivery, the potential impact on drug discovery is substantial.